 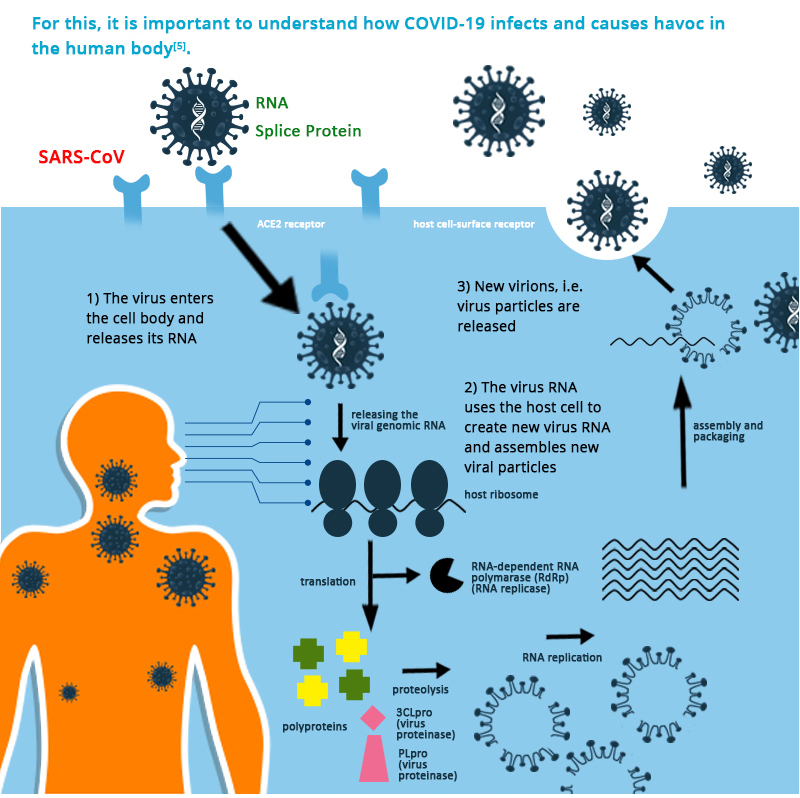 
Such functions are expected to be effective against the respiratory infections discussed here 7.Ĭonsidering the limited availability of effective therapies for COVID-19 and one of its complications ARDS, new therapeutic approaches are urgently needed. These soluble factors can promote alveolar macrophage phagocytosis and alter the cytokine profile released by immune cells. Specifically, MSCs secrete keratinocyte growth factor, prostaglandin E2, granulocyte-macrophage colony-stimulating factor, and cytokines such as IL-6 and IL-13, that can influence how immune cells-both innate and adaptive-interact with the cellular environment. These therapies have multiple therapeutic targets because MSCs can release a variety of soluble mediators, but their safety and potential efficacy are still to be determined 6. More recently, a significant number of cell-based therapies have been through clinical investigation, involving, most importantly, mesenchymal stem cells (MSCs) and MSC-derived conditioned media or extracellular vesicles. Novel therapeutic approaches have been developed to treat COVID-19 complications, especially ARDS, some of which exploit antiviral and immune-based mechanisms 5. Despite global efforts to establish effective interventions for COVID-19, its treatment remains mainly supportive, and one of the major complications of the disease, acute respiratory distress syndrome (ARDS), poses a significant challenge 3.Ĭoronaviruses that are responsible for severe respiratory syndrome (SARS) and the Middle East respiratory syndrome (MERS) belong to the same beta coronavirus genus, and SARS and MERS resemble SARS-CoV-2 symptoms 4. The virus responsible for coronavirus disease 2019 (COVID-19) infected more than 128.7 million people around the world by March 2021 2. 
In January 2020, to raise awareness internationally and to prevent further viral spread, the World Health Organization (WHO) declared the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) outbreak a public health emergency 1. Therefore, MSCs treatment could be considered a potential candidate for adjuvant therapy in moderate-to-severe COVID-19 cases and compassionate use. In addition to standard care treatments concerning COVID-19 management, there has been supportive evidence towards adjuvant therapies to reduce mortality rates and improve recovery of care treatment. Additionally, among the studies, there were no reported deaths causally linked to cell therapy. Findings from the analyzed articles indicate a positive impact of stem cell therapy on crucial immunological and inflammatory processes that lead to lung injury in COVID-19 and ARDS patients. Most of these (75%) used mesenchymal stem cells (MSCs) to treat COVID-19 and ARDS. Among the clinical studies, 21 studies used cell therapy to treat COVID-19, while eight studies investigated cell therapy as a treatment for ARDS. 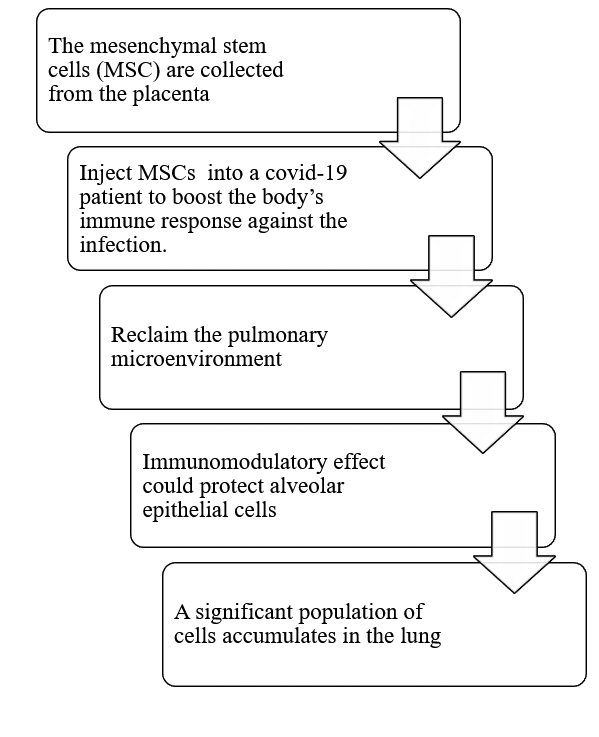
Among the 29 studies, there were eight case reports, five Phase I clinical trials, four pilot studies, two Phase II clinical trials, one cohort, and one case series. The literature search was performed using PubMed, Embase, and Scopus. In this study, we performed a systematic review of clinical studies investigating different types of stem cells as treatments for COVID-19 and ARDS to evaluate the safety and potential efficacy of cell therapy. A significant number of cell-based therapies have been through clinical investigation. Hence, identifying an effective and safe therapy for severe COVID-19 is critical for saving lives. Despite global efforts to establish effective interventions for coronavirus disease 2019 (COVID-19) and its major complications, such as acute respiratory distress syndrome (ARDS), the treatment remains mainly supportive. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
